














weaker base than HSO 4-, a consequence of the fact that its conjugate acid, HNO3, is a stronger acid than H 2SO 4.However, nitrate is not so weak that it cannot be protonated in sulfuric acid, so NO 3-is of directly measurable base strength in liquid H2SO 4.On the other hand, ClO Orthosilicate | O4Si-4 | CID 104812 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety Conjugate Acid-Base Pairs.Acids and bases exist as conjugate acid-base pairs.The term conjugate comes from the Latin stems meaning "joined together" and refers to things that are joined, particularly in pairs, such as Brnsted acids and bases..Every time a Brnsted acid acts as an H +-ion donor, it forms a conjugate base.Imagine a generic acid, HA.When this acid donates an H + ion to water Question: 7. Identify The Conjugate Bases Corresponding To The Following Acids: [CO(NH2),(OH,)", HCOCH,OH, HPO4. Si(OH)4, And H S. Also Write The Acid-base Reaction With Water For Each Acid And Label The Acid, Base, And Conjugate Acid. TABLE OF CONJUGATE ACID-BASE PAIRS Acid Base K a (25 oC) HClO 4 ClO 4 – H 2 SO 4 HSO 4 – HCl Cl– HNO 3 NO 3 – H 3 O + H 2 O H 2 CrO 4 HCrO 4 – 1.8 x 10–1 H 2 C 2 O 4 (oxalic acid) HC 2 O 4 – 5.90 x 10–2 [H 2 SO 3] = SO 2 (aq) + H2 O HSO Chemistry 401 Intermediate Inorganic Chemistry University of Rhode Island Practice Problems Acids & Bases. 1. Identify the conjugate bases corresponding to the following acids: [Co(NH 3) 5 (OH 2)] 3+, HSO 4 –, CH 3 OH, H 2 PO 4 –, Si(OH) 4, HS –.Also write the acid-base reaction with water for each acid and label the acid, base, and conjugate acid. Chemistry 401 Intermediate Inorganic Chemistry University of Rhode Island Practice Problems Acids & Bases. 1. Identify the conjugate bases corresponding to the following acids: [Co(NH 3) 5 (OH 2)] 3+, HSO 4 –, CH 3 OH, H 2 PO 4 –, Si(OH) 4, HS –.Also write the acid-base reaction with water for each acid and label the acid, base, and conjugate acid. Conjugate acids and bases are part of the Bronsted-Lowry theory of acids and bases. According to this theory, the species that donates a hydrogen cation or proton in a reaction is a conjugate acid, while the remaining portion or the one that accepts a proton or hydrogen is the conjugate base. The conjugate base may be recognized as an anion. The conjugate base is SiO(OH) 3. Discuss. A conjugate acid is a species with one more proton than the parent base. At stp, which substance is the best conductor of electricity? Since the water molecules formed as the conjugate acid of OH-have very little tendency to donate protons, they are only very weak acids. Identify the conjugate bases corresponding to the following acids: \\left[\\mathrm{Co}\\left(\\mathrm{NH}_{3}\\right)_{5}\\left(\\mathrm{OH}_{2}\\right)\\right]^{3
[index] [2810] [989] [7685] [1865] [987] [4079] [4978] [7122] [8834] [8811]
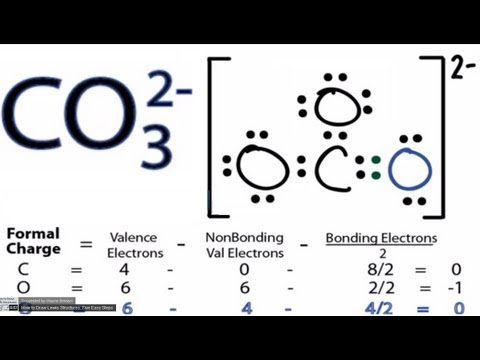
This chemistry video tutorial shows you how to identify an ionic compound as acidic, basic, or a neutral salt. You need to know the 6 common strong acids su... This channel focuses on providing tutorial videos on organic chemistry, general chemistry, physics, algebra, trigonometry, precalculus, and calculus. In this video we'll balance the equation HCl + Ca(OH)2 = CaCl2 + H2O and provide the correct coefficients for each compound. To balance HCl + Ca(OH)2 = CaCl... For early access to new videos and other perks: https://www.patreon.com/welchlabsWant to learn more or teach this series? Check out the Imaginary Numbers are... Naming acids just got a whole lot easier and one might even say fun! This video explains how to name the two types of acids, and you learn some helpful ways ... 069 - pH and BuffersIn this video Paul Andersen explains how buffer solutions maintain pH in a solution. A buffer solution is made up of a weak acid and its... This organic chemistry video tutorial provides a basic introduction into lewis acids and bases. It explains how to predict the products of a lewis acid-base... A step-by-step explanation of how to draw the CO3 2- Lewis Dot Structure (Carbonate ion).For the CO3 2- structure use the periodic table to find the total nu... This chemistry video tutorial explains how to calculate the pH of a buffer solution using the henderson hasselbalch equation. It explains the concept, compo... If you're in high school or college taking Chemistry, I can help you understand everything you need to know so you can pass! On this YouTube channel, you'll find video tutorials on all of the ...
Copyright © 2024 m.topcasinoplay.shop